“≈ќ–≈“»„≈— јя „ј—“№
–аствор- это однородна€ система, состо€ща€ из растворител€,растворенных веществ и продуктов их взаимодействи€. –астворителем чаще всего €вл€етс€ то вещество, которое в чистом виде имеет тоже агрегатное состо€ние, что и раствор, либо присутствует в избытке.
–астворителем €вл€етс€ то вещество, которое в чистом виде имеет то же агрегатное состо€ние, что и раствор, находитс€ визбытке.
ѕо агрегатному состо€нию различают растворы: жидкие, твердые, газообразные. †††††††††††††††††††††††††††††† ѕо соотношению растворител€ и растворенного вещества все растворы дел€тс€ на:
¾ Ќенасыщенный раствор Ч раствор, в котором концентраци€ растворенного вещества меньше, чем в насыщенном растворе, и в котором при данных услови€х можно растворить ещЄ некоторое его количество.
¾ Ќасыщенный раствор Ч раствор, в котором растворЄнное вещество при данных услови€х достигло максимальной концентрации и больше не раствор€етс€. ќсадок данного вещества находитс€ в равновесном состо€нии с веществом в растворе.
¾ ѕересыщенный раствор Ч раствор, содержащий при данных услови€х больше растворЄнного вещества, чем в насыщенном растворе, избыток вещества легко выпадает в осадок. ќбычно пересыщенный раствор получают охлаждением раствора, насыщенного при более высокой температуре (пересыщение).
—остав раствора обычно передаетс€ содержанием в нем растворимого вещества в виде массовой доли, процентной концентраций и мол€рности.
Ј ћассова€ дол€ ( безразмерна€ величина) Ц это отношение массы растворенного
вещества к массе всего раствора: †††††††
†††††††††††††† W м.д. = mраст. вещества
†††††††††††††††††††††††††††††††††††††††††††††††††††† m раствора.
Ј ѕроцентна€ концентраци€ (%) Ц это величина показывающа€ сколько грамм† растворенного вещества cсодержитс€ в 100 гр. раствора :††††††††††††††††††††
††††††††††††††††††††††††††††††† W % =†††† mраст. вещества 100%
††††††††††††††††† †††††††††††††††††††††††††††††††††††††††† m раствора
Ј ћол€рна€ концентраци€, или мол€рность (моль/литр)- это величина показывающа€ сколько молей растворимого вещества содержатс€ в 1 литре раствора:††††††††††††††††††††††††††††††††
†††††††††††††† †††††††††††††††††† —м =†††† mраст. вещес
††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††††† ћ r (раст. вещества ) V раствора .
ѕ–ј “»„≈— јя „ј—“№
| ’од работы, задание | –ешение, расчеты | |||
| ќпыт є1 ѕ–»√ќ“ќ¬Ћ≈Ќ»≈ –ј—“¬ќ–ј ѕќ¬ј–≈ЌЌќ… —ќЋ»† | ||||
| «адание: †ќпределить процентную и мол€рную концентрацию раствора массой 20 гр., содержащего 3гр. соли NaCL, плотность которого 1,2 гр/л | ƒано: | –ешение: ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ
| ||
| Ќайти: | ||||
| ќпыт є2 † †† ѕ–»√ќ“ќ¬Ћ≈Ќ»≈ —ј’ј–Ќќ√ќ –ј—“¬ќ–ј –ј«Ѕј¬Ћ≈Ќ»≈ћ | ||||
| «адание: ќпределить массу воды и растворЄнного в ней сахара, если масса раствора 150гр., а процентна€† концентраци€ сахара в растворе равна 40%. ак изменитс€ концентраци€ раствора если к нему прилить 30гр. воды | ƒано: ............................................................................................................................................................ †Ќайти ....................................................† | –ешение: ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ | ||
| ќпыт є3 ѕ–»√ќ“ќ¬Ћ≈Ќ»≈ —ј’ј–Ќќ√ќ –ј—“¬ќ–ј — ЅќЋ№Ў≈… ќЌ÷≈Ќ“–ј÷»≈…
| ||||
| ќпределить массу раствора сахара в воде, если масса сахара в нЄм 15гр., а процентна€† концентраци€ 45%. ак изменитс€ концентраци€ раствора,если к нему добавить 5гр.сахара | ƒано: ............................................................................................................................................................ †Ќайти ....................................................† | –ешение: ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ | ||
| ќпыт є4 † ѕ–»√ќ“ќ¬Ћ≈Ќ»≈ –ј—“¬ќ–ј ” —”—Ќќ… »—Ћќ“џ »« 2-’ –ј—“¬ќ–ќ¬ | ||||
| —мешали 40 гр. 9%-ного раствора уксуса и 20гр. 70%-ного раствора уксусной эсенцинции. ќпределить концентрацию полученного раствора уксусной кислоты. | ƒано: ............................................................................................................................................................ .................................................... .................................................... †Ќайти ....................................................† | –ешение: ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ | ||
| ќпыт є5 † †† ѕ–»√ќ“ќ¬Ћ≈Ќ»≈ –ј—“¬ќ–ј «јƒјЌЌќ… ќЌ÷≈Ќ“–ј÷»» | ||||
| —колько грамм сахара необходимо добавить к 300г. 60%-ного сахарного раствора, чтобы он стал 90%-ным | ƒано: ............................................................................................................................................................ †Ќайти ....................................................† | ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ | ||
ќбщий вывод: ............................................................................................................................................................................................
ЕЕЕЕЕЕЕЕ............................................................................................................................................................................ЕЕЕЕЕЕЕЕ...........................................................................................................................................................................................................
ЕЕЕЕ...........................................................................................................................................................................................................
ЕЕЕЕ...........................................................................................................................................................................................................
|
|
|
ЋјЅќ–ј“ќ–Ќјя –јЅќ“ј є3
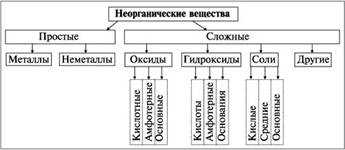
“ема: " лассы неорганических веществ"
÷ель: ѕознакомитьс€ с основными классами неорганических веществ, †исследовать наиболее распространенные свойства данных соединений
“≈ќ–≈“»„≈— јя „ј—“№
ѕ–ј “»„≈— јя „ј—“№
| ’од работы | –исунки, реакции, наблюдени€. | ¬ыводы |
| ќпыт є 1 ќ«Ќј ќћЋ≈Ќ»≈ —ќ —¬ќ…—“¬јћ» »—Ћќ“ ислоты Ц сложные вещества, содержащие в своем составе водород и кислотныей остаток. | ||
| 1.1 ћен€ют цвет индикатора ¬ 3-пробирки влить по 1мл. Ќ—L и добавим лакмус, метилоранж, фенолфталеина. | Ќаблюдали: ............................................................................ ........................................................................................................................................................ | |
| 1.2 ¬заимодействуют с металлами a) ¬ 2-пробирки влить по 1-2мл. сол€ной кислоты и добавим кусочки меди и цинка. b) ¬ 2 пробирки влить 1-2мл разбавленной азотной кислоты и добавить туда кусочки меди и цинка. ¬ 3 пробирки влить 1-2мл концентрированной азотной кислоты и добавить туда кусочки меди, цинка и алюмини€ c) ¬ 2 пробирки влить 1-2мл разбавленной серной кислоты и добавить туда кусочки меди и цинка. ¬ 2 пробирки влить 1-2мл концентрированной серной †кислоты и добавить туда кусочки меди, цинка. | ј) ƒопишем реакции: HCL +Zn→....................................................... HCL +Cu→....................................................... ............................................................................ ............................................................................ Ѕ) ƒопишем реакции: HNO3(разб) +Zn→≠NH3 +................................... HNO3(разб) +Cu→ ≠Nќ +.................................. HNO3(конц) +Zn→≠Nќ2 +.................................. HNO3(конц) +Cu→≠ Nќ2 +................................ HNO3(конц) +Al→.............................................. ............................................................................ ............................................................................ ............................................................................ .......................................................................... —) ƒопишем реакции: H2 SO4(разб) +Zn→≠H2+..................................... H2 SO4(разб) +Cu→........................................... H2 SO4(конц) +Zn→≠Sќ2 +............................... H2 SO4(конц) +Cu→≠ Sќ2 +............................... ............................................................................ ............................................................................ ............................................................................ | |
| 1.3. ¬заимодействуют с основными оксидами ¬ 3-пробирки влить по 1мл. Ќ—L и добавим оксиды Fe2O3 и Cаќ. ѕробирку с †Fe2O3 нагреем | ƒопишем реакции: HCL +Fe2O3→................................................... HCL +Cаќ→.................................................... Ќаблюдали: ............................................................................ ............................................................................ | |
| 1.4. ¬заимодействуют с сол€ми ¬ пробирку влить 1мл серной кислоты и добавить хлорид бари€. Ќанести 1мл сол€ной кислоты на кусок мрамора. | ƒопишем реакции: HCL +—а—ќ3 →............................................... H2 SO4+¬а—L2 →............................................. Ќаблюдали: ............................................................................ ............................................................................ ...................................................................................................................................................... | |||
| ќпыт є 2 ќ«Ќј ќћЋ≈Ќ»≈ —ќ —¬ќ…—“¬јћ» ќ—Ќќ¬јЌ»…
ќсновани€ -это соединени€, состо€щие из катионов металла (или катиона аммони€) и гидроксильной группы ќЌ1- | ||||
| 2.1 ћен€ют цвет индикатора ¬ 3-пробирки влить по 1мл. NaOH и добавим лакмус, метилоранж, фенолфталеина | Ќаблюдали: .................................................................................................................................................................................................................. | |||
| 2.2 ¬заимодействуют с кислотами, вступа€ в реакцию нейтрализации ¬ пробирку влить по 1мл. NaOH и добавим пару капель фенолфталеина, после чего добавить HCL | ƒопишем реакцию: NaOH +HCL→.................................................. Ќаблюдали: ............................................................................ ............................................................................ ........................................................................... | |||
| 2.3¬заимодействуют с сол€ми ¬ пробирку поместим 1мл раствора CuSO4, добавим по 1-2 капли NaOH и индикатора. ќсадок меди будем длительно нагревать до его разложен舆 | ƒопишем реакцию: —uSO4+ NaOH Ѓ ЕЕЕЕЕЕЕЕЕЕ —u(ќЌ)2 нагреем ЃЕЕЕЕЕЕ............ Ќаблюдали: ............................................................................ ............................................................................ | |||
| ќпыт є3 ќ«Ќј ќћЋ≈Ќ»≈ —ќ —¬ќ…—“¬јћ» —ќЋ≈… —оли Ц это продукты полного или частичного замещени€ атомов ЂЌї в молекуле кислоты на металл(либоNH4+), либо групп ЂќЌї в молекуле основани€ на кислотный остаток | ||||
| 3.1 ¬заимодействуют друг с другом ¬ пробирку поместим 1мл. раствора сульфида натри€ Na2S, добавим 1мл. нитрата серебра AgNO3. | ƒопишем реакцию: Na2S+ AgNO3Ѓ ЕЕЕЕЕЕЕЕЕЕ Ќаблюдали: ............................................................................ ............................................................................ | |||
| 3.1 ¬заимодействуют c †металлами ¬ 3пробирки †поместим по 1мл. растворов †AgNO3, —uSO4 Pb(NO3)2добавим металлы соответственно †††—u, †Fe, Zn | ƒопишем реакцию: —u+ AgNO3Ѓ ЕЕЕЕЕЕЕЕЕЕ..Е Е Fe + —uSO4 Ѓ ЕЕЕЕЕЕЕЕЕЕЕЕ Zn+ Pb(NO3)2Ѓ ЕЕЕЕЕЕЕЕЕЕЕ.. Ќаблюдали: ............................................................................ ........................................................................... ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ... | |||
ќбщий вывод: .............................................................................................................................................................................................
ЕЕЕЕЕЕЕЕ............................................................................................................................................................................ЕЕЕЕЕЕЕЕ............................................................................................................................................................................ЕЕЕЕЕЕЕЕ....................................................................................................................................................................................................
ЋјЅќ–ј“ќ–Ќјя –јЅќ“ є4
“ема: †††† "√идролиз солей, испытание кислот и щелочей индикаторами"
÷ель: ѕознакомитьс€ с пон€тием Ђгидролиз солейї.Ќаучитьс€ определ€ть среду раствора солей, кислот, щелочей с помощью индикаторов и составл€ть реакции гидролиза.
“≈ќ–≈“»„≈— »… ќЅ«ќ–
√идролиз Ц это процесс взаимодействи€ ионов соли с водой, привод€щий к образованию слабого электролита.†
|
|
|
¬се соли можно разделить на 4 группы:
1. —оль образована сильным основанием и сильной кислотой 2 Sќ4, Na NO3,)Ц гидролиз не идет, среда нейтральна€ рЌ = 7.
2. —оль образована слабым основанием и слабой кислотой (Mg—ќ3, Al 2S3, Zn(NO2)2, †..) - гидролиз протекает практически в нейтральной среде рЌ ближе к 7, гидролиз идет по катиону и аниону:†††
3. —оль образована сильным основанием и слабой кислотой (например: Na2—ќ3, 2S, ¬а(NO2)2, —Ќ3—ќќ Li) -гидролиз протекает в щелочной среде рЌ >7, гидролиз идет по аниону.
4. —оль образована слабым основанием и сильной кислотой (MgSќ4, AlCL3, Zn(NO3)2, †..) - гидролиз протекает в кислой среде рЌ< 7, гидролиз идет по катиону.
√лубина гидролиза зависит от температуры (чаще всего ее приходитс€ повышать) и концентрации раствора (при разбавлении раствора гидролиз усиливаетс€)
≈сли продукты гидролиза летучи,или нерастворимы, то он необратим.
—ведени€ по веществам
Ј —ильные кислоты Ц кислоты, €вл€ющиес€ сильными электролитами (H2SO4, HCl, HNO3, HBr, HI, HClO4 и другие).
Ј —лабые кислоты Ц кислоты, €вл€ющиес€ слабыми электролитами (H2CO3, H2SO3, H2S, H2SiO3, H3PO4, Ќ—N и другие)
Ј —ильные основани€ Ц сильные электролиты - щелочи (NaOH, KOH, и другие).
Ј —лабые основани€ Ц нерастворимые основани€, слабые электролиты (Cu(OH)2, Ca(OH)2, NH4OH, Al(OH)3 и другие.
’ќƒ –јЅќ“џ
| ’од работы | –исунки, реакции, наблюдени€ | ¬ыводы | ||
| ќпыт є1 ќпределение характера среды кислот и щелочей | ||||
| ¬озьмЄм по 3 †стекл€нных пробирки и заполним их сол€ной кислотой и щЄлочью натри€ ƒобавим в них индикаторы: лакмус, фенолфталеин и метилоранж . |
ЌCL (рЌ<7)††††††††††††††††††††††† NaOЌ(рЌ>7)
   †††††††††††† ††††††††††††    Ќаблюдали:
ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ..
ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ
Ќаблюдали:
ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ..
ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ
| ............................... ............................... ................................ ................................ ................................. | ||
| ќпыт є2 ќпределение характера среды растворов солей, кислот, щелочей. | ||||
| †ј) √идролиз соли,образованной сильными основание и кислотой ¬ пробирку нальем 1 мл раствора Na2SO4,- внесем- 2-капли индикатора-лакмуса | Ќаблюдали: ........................................................................................................................................................................................................................................................................................................... —оставить уравнени€ гидролиза: Na2SO4+ ЌќЌ→................................................... ............................................................................................................................................................... | |||
| б) √идролиз соли,образованной слабой кислотой и сильным основанием † ¬ пробирку нальем 1 мл раствора соли Na2—ќ3 и внесем 2 капли лакмуса. | Ќаблюдали:
 ...........................................................................................................................................................................................................................................................
—оставить уравнени€ гидролиза:
Na2CO3+ ЌќЌ →....................................
............................................................................
.............................................................................
Nа2SiO3+ ЌќЌ →......................................
............................................................................
............................................................................. ...........................................................................................................................................................................................................................................................
—оставить уравнени€ гидролиза:
Na2CO3+ ЌќЌ →....................................
............................................................................
.............................................................................
Nа2SiO3+ ЌќЌ →......................................
............................................................................
.............................................................................
| |||
| в) √идролиз соли, образованной слабым основанием и сильной кислотой. ¬ пробирку нальем 1 мл раствора соли ZnSO 4 и внесем 1-2 капли лакмуса ¬ пробирку нальем 1 мл раствора соли CuSO 4 и внесем 1-2 капли лакмуса | Ќаблюдали:
 ...........................................................................................................................................................................................................................................................................................................
—оставить уравнени€ гидролиза:
ZnSO4+ ЌќЌ Ѓ................................................
..............................................................................................................................................................
—uSO4+ ЌќЌ Ѓ....................................................
................................................................................
.............................................................................................................................................................. ...........................................................................................................................................................................................................................................................................................................
—оставить уравнени€ гидролиза:
ZnSO4+ ЌќЌ Ѓ................................................
..............................................................................................................................................................
—uSO4+ ЌќЌ Ѓ....................................................
................................................................................
..............................................................................................................................................................
| |||
| ќпыт є3 †¬ли€ние температуры на гидролиз
| ||||
| ¬ пробирку нальем 1 мл раствора соли Na2SO3 и 1мл фенолфталеина. †ѕоместим в раствор индикаторную бумагу, нагреем содержимое пробирки в пламени горелки.† | Ќаблюдали: ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ.. Na2SO3+ ЌќЌ Ѓ................................................ ................................................................................. ................................................................................................................................................................................................................................................................................................................................................................................................................. | |||
ќбщий вывод:ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ
ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ.................
ЋјЅќ–ј“ќ–Ќјя –јЅќ“ј є5
“ема: †††† " –еакции соединени€, разложени€, замещени€, обмена, эндо-и экзо-термические реакции. –»ќ, идущие необратимо"
÷ель: Ќаучитьс€ практически выполн€ть реакции соединени€, разложени€, замещени€, ионного обмена. составл€ть –»ќ, идущие необратимо.
“≈ќ–≈“»„≈— »… ќЅ«ќ–
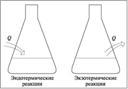
Ј Ёкзотермическа€ реакци€ Ч химическа€ реакци€, сопровождающа€с€ выделением тепла.
Ј Ёндотермическа€ реакци€ Ч химическа€ реакци€, сопровождающа€с€ поглощением тепла.
Ј –еакци€ замещени€ Ч это реакции, в результате которых происходит замещение одних атомов, содержащихс€ в молекуле, на другие. ¬ данную реакцию вступает одно сложное вещество и одно простое, в результате образуетс€ новое простое и новое сложное вещества:—uSO4+ ZnЃZnSO4+ Cu ††
Ј –еакци€ разложени€ Ч это реакции, в результате которых из исходного вещества образуетс€ два и более новых веществ: —а—ќ3 нагревание Ѓ —аќ+—ќ2≠
Ј –еакции обмена Ч это реакции, в результате которых происходит обмен атомами, вход€щими в состав молекулы: ЌCL+AgNO3Ѓ AgCL¯+ HNO3
Ј –еакци€ присоединени€ †Ч это реакции в результате которых из исходных веществ получаетс€ одно новое вещество: Ќ2+ CL2Ѓ2HCL.
»онные реакции -реакции †протекающие в растворах между ионами.
–еакции ионного обмена не сопровождаетс€ изменением зар€да ионов (степени окислени€ атомов). ¬озможно †два варианта исхода –»ќ: †††а) реакци€ идет обратимо; б) реакци€ идет необратимо.
–еакции ионного обмена идут в соответствии со схемой: AB + C ƒ à јƒ + ¬—
–»ќ протекает необратимо в трех случа€х, когда образуетс€: трудно растворимое †соединение(осадок), газообразные †вещества †(H2S, CO2, NH3...), малодиссоциирующих веществ (Ќ2ќ, Ќ—N,CH3COOH, ЌNO2, H3PO4)
Ќестойкие соединени€. NH4OH→ NH3 ≠ + H2O; H2CO3 → CO2 ≠ + H2O; H2SO3 → H2O + SO2 ≠
’ќƒ –јЅќ“џ
| ’од работы | –исунки, реакции, наблюдени€. | ¬ыводы | |||||
| ќпыт є 2 †–≈ј ÷»» ѕ–»—ќ≈ƒ»Ќ≈Ќ»я, –ј«Ћќ∆≈Ќ»я » «јћ≈ў≈Ќ»я | |||||||
| а)–еакци€ замещени€ ¬ пробирку † поместим 1мл. Cu SO4 и железную скрепку (внесем в пробирку 1-2 капли Ќ2SO4 раз.) красной кров€ной соли. | —оставим уравнени€ реакций: а) Cu SO4 + Fe ЃЕ..ЕЕЕЕЕЕЕЕЕЕ
ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕЕЕЕЕ.... |
ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ. | |||||
| б)–еакци€ разложени€ ¬ фарфоровую чашку поместить небольшой горкой дихромат аммони€ (NH4)2Cr2O7 †и ввести в центр горки гор€щую спичку. | Ќаблюдали: | ||||||
| (NH4)2Cr2O7 †ЃN2 + Cr2O3 + 4H2O
ЕЕЕЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕЕЕ
| |||||||
| в) –еакци€ присоединени€ ∆елезную скрепку внесем в плам€ горелки (образуетс€ оксид Fe2ќ3) | |||||||
| в) O2 + Fe ЃЕ..ЕЕЕЕЕЕЕЕЕЕЕ Ќаблюдали: ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ........ | |||||||
|
ќпыт є3 –≈ј ÷»» »ќЌЌќ√ќ ќЅћ≈Ќј »ƒ”ў»≈ Ќ≈ќЅ–ј“»ћќ | |||||||
| а)ќбразование осадка. ¬ пробирку к 1 мл раствора сульфата меди ††прильем по капл€м раствор щелочи | —оставим –»ќ: —uSO4+ NaOHЃЕЕЕЕЕЕЕЕЕЕЕЕ .................................................................................... ............................................................................... Ќаблюдали: |
ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ. | |||||
|
| .................................................................................................................................................. | ||||||
| б)ќбразование газа ¬ пробирку с 1 мл раствора карбоната натри€ (Na2CO3) осторожно прильем 1 мл сол€ной кислоты | HCL + Na2CO3ЃЕЕЕЕЕЕЕЕЕЕЕЕ .................................................................................... ...................................................................................... Ќаблюдали: | ||||||
|
| .............................................................................................................................................................................................................. | ||||||
| в)ќбразование воды ¬ пробирку к 1 мл раствора едкого натра прильем каплю фенол-фталеина и добавим 1 мл.. | NaOH + HCL Ѓ.................................................. ...............ЕЕЕЕ.ЕЕЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕЕЕЕЕ........................... Ќаблюдали:.................................................................................... ...................................................................................... | ||||||
| г)ќбразование осадка и его растворение ¬ пробирке смешаем по 1 мл раствора cжелеза (III) и едкого натра. полученному осадку гидроксида железа(III) прильем раствор †HCL | FeCl3 + NaOHЃ ЕЕЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ Fe(OH)3 + HCL Ѓ ЕЕЕЕЕЕЕ .................................................................................................................................................................... Ќаблюдали: | ||||||
|
| |||||||
| ќпыт є3 –ј—ѕќ«Ќќ¬јЌ»≈ Ё «ќ“≈–ћ»„≈— »’ » ЁЌƒќ“≈–ћ»„≈— »’ –≈ј ÷»» | |||||||
| а)¬ стекл€нный стакан внести 2-3 гранулы NaOH и добавим воду. »змерим температуру раствора термометром. † | Ќаблюдали:.................................................................................. .................................................................................. .................................................................................. ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ..
|
................................................................................................................................................................................................. | |||||
| б) ¬ пробирку к 1 мл раствора NaOH добавим 2-4 капли раствора NH4CL, нагреем до по€влени€ запаха | NH4CL+NaOH→.....................................................ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕЕЕЕЕ............... Ќаблюдали:..................................................................................
| ||||||
ќбщий вывод: ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ
ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ
ЋјЅќ–ј“ќ–Ќјя –јЅќ“ј є6
“ема: "‘акторы, вли€ющие на скорость реакции."
÷ель: ѕознакомитьс€ с пон€ти€ми: гомогенна€ и гетерогенна€ реакци€, скорость реакции, химическое равновесие. Ќаучитьс€ составл€ть уравнени€ пр€мой и обратной реакции, выражать константу равновеси€, определ€ть зависимость скорости от природы реагирующих веществ, их концентрации, температуры, поверхности соприкосновени€ и использовани€ катализаторов.
“≈ќ–≈“»„≈— »… ќЅ«ќ–
—корость химической реакции ( ט )- определ€етс€ изменением концентрации одного из реагирующих веществ в единицу времени: ט = ∆— / ∆ t †††††††††††††††††††††††††††† ††††††
Ј √омогенные системы - реакции, компоненты которых наход€тс€ в одинаковом физическом состо€нии -в газообразном или растворенном:
ајгаз + в¬газ→с—газ + dD газ
ѕо закону действующих масс:
-—корость пр€мой реакции равна:† ט †пр. = пр. ∙ —ј а ∙ —¬ в = пр
-—корость обратной реакции равна: ††ט обр.= обр.. ∙ —— с ∙ —D d= обр..
ü - константа скорости, величина посто€нна€;†
ü — (–)- мол€рные концентрации(давление) реагентов
Ј √етерогенные системы - реакции, компоненты которых наход€тс€ в разных физических состо€ни€х -в газообразном, в растворенном, в твердом.
—корость реакции измен€етс€ только в зависимости от концентрации газов и растворенных веществ:†††
ајтверд.. + в¬газ→с—жидк.
-—корость пр€мой реакции равна:† ט †пр. = пр. ∙ —¬ в
-—корость обратной реакции равна: ††ט обр.= обр.. ∙ —— с
‘ а к т о р ы, в л и € ю щ и е н а с к о р о с т ь р е а к ц и и.
- ѕрирода реагирующих веществ
- онцентраци€ реагирующих веществ.
„ем выше концентраци€ веществ, тем больше скорость, протекающей реакции.
- “емпература - зависимость скорости реакции от температуры выражаетс€ правилом ¬ант
√оффа: при повышении температуры на 10 градусов скорость химической реакции увеличиваетс€ в 2-4 раза, где V 2 и V 1 - скорости реакций при температурах t 2 и t 1; γ - температурный коэффициент скорости реакции.
- ѕоверхность реагирующих веществ.
- »нгибитор, катализатор.
»зменение скорости реакции под действием катализаторов называют - катализом.
’имическое равновесие - это такое состо€ние обратимого химического процесса, при котором скорости пр€мой и обратной реакций равны между собой: ט пр. = ט † обр†††††††††††† онцентрации реагирующих веществ, которые устанавливаютс€ при химическом равновесии, называютс€ равновесными.
ѕ–ј “»„≈— јя „ј—“№
| ’од работы | –исунки, реакции, наблюдени€. | ¬ыводы | |
| †ќпыт є «ј¬»—»ћќ—“№ — ќ–ќ—“» –≈ј ÷»» ќ“ ќЌ÷≈Ќ“–ј÷»» | |||
| а) ƒобавим к цинку серную кислоту в 2х различных концентраци€х. ѕо количеству выделенного Ќ2 определим, как вли€ет концентраци€ кислоты на скорость реакции б) ак изменитс€ скорость реакции, если увеличить концентрацию Ќ2 в 2раза | а)Zn+ Ќ2SO4Ѓ ЕЕЕЕЕЕЕЕЕЕЕЕЕЕ ” равнени€ скорости пр€мой и обратной реакции: ........................................................................................................................................................................ Ќаблюдали: ЕЕ............................................................................. ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ б) N2(г)+ 3H2 (г) →2NH3(г) –асчет: ЕЕ............................................................................. ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ ЕЕ............................................................................. | ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ | |
|
ќпыт є2 «ј¬»—»ћќ—“№ — ќ–ќ—“» –≈ј ÷»» ќ“ “≈ћѕ≈–ј“”–џ | |||
| а) ƒобавим к цинку серную кислоту при 2х различных температурах. ѕо количеству выделенного Ќ2 определим, как вли€ет температура кислоты на скорость реакции б) ќпределить скорость реакции при 600—, если при 200— скорость равна 0,5моль/л.с., а температурный коэффициент- 2 | а)Ќаблюдали:
ЕЕ..................................................................................................................................................................................................................................................
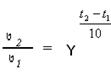 б)–асчет:
ЕЕ.....................................................................................................................................................................................................................................................................................................................................
б)–асчет:
ЕЕ.....................................................................................................................................................................................................................................................................................................................................
| ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ | |
| ќпыт є3 «ј¬»—»ћќ—“№ — ќ–ќ—“» –≈ј ÷»» ќ“ ѕќ¬≈–’Ќќ—“» –≈ј√»–”ёў»’ ¬≈ў≈—“¬ | |||
| ƒобавим к гранулам цинка различного размера серную кислоту. ѕо количеству выделенного Ќ2 определим, как вли€ет поверхность веществ на скорость реакции. | † Ќаблюдали: ЕЕ.............................................................................................................................................................................................................................................. .................................................................................................................................................................... | ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ | |
| ќпыт є4 «ј¬»—»ћќ—“№ — ќ–ќ—“» –≈ј ÷»» ќ“ ѕ–»–ќƒџ –≈ј√»–”ёў»’ ¬≈ў≈—“¬ | |||
| ƒобавим †к гранулам цинка †уксусную и серную кислоты. ѕо количеству выделенного Ќ2 определим вли€ние природа веществ на скорость реакции. | † —оставим реакции: —Ќ3—ќќЌ + Zn→ЕЕЕЕЕЕЕЕЕЕЕЕ H2SO4+ Zn→ЕЕЕЕЕЕЕЕЕЕЕЕ Ќаблюдали: ЕЕ............................................................................................................................................................... | ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ | |
| ќпыт є5 «ј¬»—»ћќ—“№ — ќ–ќ—“» –≈ј ÷»» ќ“ ј“јЋ»«ј“ќ–ј » »Ќ√»Ѕ»“ќ–ј | |||
| ј) —равним разложение перекиси водорода †в присутствии ћnO2 †и без него. Ѕ) —равним количество образованного Ќ2 при взаимодействии цинка с H2SO4 в присутствии формалина и без него. | —оставим реакции: 2Ќ2ќ2 MnO 2 → 2Ќ2ќ †+ †ќ2 H2SO4+ Zn→ЕЕЕЕЕЕЕЕЕЕЕЕ Ќаблюдали: ЕЕ......................................................................................................................................................................................................................................................... | ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ | |
| ќпыт є6 ’»ћ»„≈— ќ≈ –ј¬Ќќ¬≈—»≈ | |||
| ¬ 3 пробирки нальЄм раствор хлорида железа и прильЄм в них родонит кали€ KCNS, растворы разбавим водой. ѕоочерЄдно добавим в растворы исходные реагенты и хлорид железа. | —оставим реакцию: FeCl3(ж)+3KCNS(ж) =Fe(CNS)3(ж)+3KCl(ж) † равнов.=
†Ќаблюдали: ЕЕ...............................................................................ЕЕ..................................................................................................................................................................... | ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ | |
ќбщий вывод:
ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ
ЋјЅќ–ј“ќ–Ќјя –јЅќ“ј є7
“ема: "ѕолучение чугуна и стали. "
÷ель: ќзнакомитс€ с процессами производства чугуна и стали с помощью флеш -анимации; изучить химические процессы производства, сырьЄ, продукты, виды чугуна, виды сталей; научитьс€ выполн€ть расчеты, св€занные с получением металлов.
“≈ќ–≈“»„≈— »… ќЅ«ќ–
„угун- сплав железа с углеродом (1,7-5%) и металлургическими примес€ми (Mn, Si, P и др.)
ƒл€ выплавки чугуна в доменных печах используют: руду: магнитный железн€к (Fe3O4); красный железн€к (Fe2O3), бурый железн€к (гидраты оксидов железа 2Fe2O3 * 3H2O и Fe2O3 * H2O) †и др.
Ј “опливо: ††кокс, †но возможна частична€ замена газом, мазутом.
Ј ‘люсы: †известн€к CaCO3, †доломитизированный известн€к(CaCO3 + MgCO3)
“≈’ЌќЋќ√»„≈— »… ѕ–ќ÷≈——
I. ѕодготовка руд ы - дробление и сортировка, обогащение руды (промывка, магнитна€ сепараци€), агломераци€. ƒл€ агломерации шихту, состо€щую из железной руды (40Е50 %), известн€ка (15Е20 %), кокса (4Е6 %), влаги (6Е9 %), спекают при температуре 1300Е1500 0—., образуютс€ окатыши.
II. 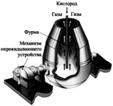 ƒоменный процесс. ј гломерат направл€ют в доменную печь, где при температуре1000Е1100 0C.восстановленное из руды твердое железо, взаимодейству€ с оксидом углерода, коксом и сажистым углеродом, интенсивно раствор€ет углерод. апли железоуглеродистого сплава, протека€ по кускам кокса, дополнительно насыщаютс€ углеродом (до 4%), марганцем, кремнием, фосфором которые при температуре 1200 0Cвосстанавливаютс€ из руды, и серой, содержащейс€ в коксе. ¬ нижней части доменной печи образуетс€ шлак в результате сплавлени€ окислов пустой породы руды, флюсов и золы топлива. †—ливают чугун каждые 3Е4 часа, шлак -1,5-2 часа. †Ўлаки содержат Al2O3, CaO, MgO, SiO2, MnO, FeO, CaS. „угун отправл€ют на производство стали †или формуют (литьем) в виде чушек-слитков массой 45 кг.
ƒоменный процесс. ј гломерат направл€ют в доменную печь, где при температуре1000Е1100 0C.восстановленное из руды твердое железо, взаимодейству€ с оксидом углерода, коксом и сажистым углеродом, интенсивно раствор€ет углерод. апли железоуглеродистого сплава, протека€ по кускам кокса, дополнительно насыщаютс€ углеродом (до 4%), марганцем, кремнием, фосфором которые при температуре 1200 0Cвосстанавливаютс€ из руды, и серой, содержащейс€ в коксе. ¬ нижней части доменной печи образуетс€ шлак в результате сплавлени€ окислов пустой породы руды, флюсов и золы топлива. †—ливают чугун каждые 3Е4 часа, шлак -1,5-2 часа. †Ўлаки содержат Al2O3, CaO, MgO, SiO2, MnO, FeO, CaS. „угун отправл€ют на производство стали †или формуют (литьем) в виде чушек-слитков массой 45 кг.
III. —таль Ц сплав железа с углеродом (до 1,7%) и металлургическими примес€ми Mn, —r, Si,Ni. ќсновными исходными материалами €вл€ютс€ передельный чугун и стальной лом (скрап). —пособы получени€ стали:
Ј онверторный способ основан на продувке сжатым воздухом расплавленного чугуна.
Ј 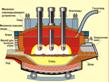 ћартеновский способ вызван к жизни необходимостью перерабатывать стальной лом
ћартеновский способ вызван к жизни необходимостью перерабатывать стальной лом
Ј ѕлавка стали в электропечах дает возможность получать высококачественные стали.
ѕо химическому составу стали дел€т на углеродистые и легированные.
¾ ”глеродистые стали, кроме углерода, содержат до 0,35% кремни€, 0,8% марганца, 0,06% серы, 0,07% фосфора. –азличают мало-, средне- и высокоуглеродистые стали.
¾ Ћегированные стали †имеют в своем составе †легирующие элементы (хром, никель, вольфрам, ванадий, молибден, кобальт и др.) дл€ сообщени€ стали требуемых свойств.
Ќержавеющей сталью называетс€ сталь, обладающа€ стойкостью против атмосферной коррозии. Ёто †обеспечиваетс€ введением легирующих элементов хрома и †меди.
ѕо назначению дел€тс€ на:
¾ онструкционные;
¾ инструментальные;
¾ стали с особыми свойствами.
ѕо способу выплавки различают сталь обыкновенного качества, качественную и высококачественную.
«акалка Ч распространенный процесс термической обработки стальных деталей. ќна осуществл€етс€ путем нагрева деталей,выдержки при этой температуре и быстрого охлаждени€. ќсновна€ цель закалки стали Ч получение высокой твердости, износостойкости и физико-механических свойств.
ќтпуск заключаетс€ в нагреве закаленной заготовки до определенной температуры и последующем охлаждении ее на воздухе, в воде, масле или других охлаждающих средах. ќтпуск уменьшает хрупкость, повышает в€зкость, улучшает †обрабатываемость резанием.
ѕ–ј “»„≈— јя „ј—“№
| ’од работы | –исунки, реакции, наблюдени€. | ¬ыводы | |
| ќпыт є 1 »——Ћ≈ƒќ¬јЌ»≈ ѕ–ќ÷≈——ј ѕќЋ”„≈Ќ»я „”√”Ќј »—“јЋ» | |||
ѕросмотрим видео анимацию. "ѕроизводство чугуна и стали ".
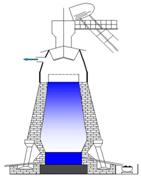 ƒоменна€ печь
ƒоменна€ печь
| ќпределим формулы соединений, дадим описание сырь€ и продуктов реакций: 1. окс.................................................................................................................. 2. »звестн€к......................................................................................................... 3. ћагнитный железн€к..................................................................................... 4. расный железн€к......................................................................................... 5. Ѕурый железн€к............................................................................................... 6. „угун................................................................................................................ 7. Ўлак................................................................................................................. 8. —таль................................................................................................................. †††††††††††††† | ||
| ќпыт є 2 »——Ћ≈ƒќ¬јЌ»≈ ќЅ–ј«÷ќ¬ —“јЋ» | |||
| –ассмотрим образцы 1. малоуглеродистую 2. среднеуглеродистую 3. высокоуглеродистую 4. нержавеющую | ќписание образцов стали: 1. ЕЕЕЕЕЕ..ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ 2. ЕЕЕЕЕ..ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ 3. ЕЕЕЕЕЕЕЕЕЕЕЕ..ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ 4. ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ | ||
| ќпыт є 3 ќѕ–≈ƒ≈Ћ≈Ќ»≈ ћј——ќ¬ќ… ƒќ» ћ≈“јЋЋј ¬ –”ƒ≈ | |||
| «адача є2 ќпределить массовую долю алюмини€ в природном соединении 2ќ* Al2O3*6SiO2 | ƒано: ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ †Ќайти ....................................................† | –ешение: ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ | |
| ќпыт є 4 ќѕ–≈ƒ≈Ћ≈Ќ»≈ ћј——џ ћ≈“јЋЋј ¬ –”ƒ≈ | |||
| «адача –уда массой 500кг, содержит 25% рутила TiO2, †определить массу титана, полученного из этой руды: | ƒано: ............................................................................................................................................Ќайти ..................................... | –ешение: ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ | |
| ќпыт є 5 ќѕ–≈ƒ≈Ћ≈Ќ»≈ ћј——џ –”ƒџ | |||
| «адача ќпределить массу руды, содержащую 20% Fe3O4, †если из неЄ получено 40г железа. | ƒано: ............................................................................................................................................ †Ќайти ....................................................† | –ешение: ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ.
| |
ќбщий вывод:...........................................................................................................................................................
...............................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................
ЋјЅќ–ј“ќ–Ќјя –јЅќ“ј є8
“ема: " ѕолучение, собирание и распознавание газов. –ешение экспериментальных задач."
“еоретический обзор
Ј ”глекислый газ или оксид углерода (IV) —ќ2 Ц бесцветный, не имеющий запах газ.
ќн примерно в полтора раза т€желее воздуха. –астворим в воде. ¬ лаборатории углекислый газ получают действием сол€ной кислоты на карбонат кальци€:
CaCO3 + 2HCl = CaCl2 + H2O + CO2↑.
–аспознание:
- ѕомутнение известковой воды (продувание углекислого газа через известковую воду)
—ќ2 + —а(ќЌ)2 = —а—ќ3 + Ќ2ќ; - √ор€щую лучину опустить в сосуд с углекислым газом. Ћучина гаснет.
Ј ¬одород (Ќ2) Ц самыйлегкий, бесцветный, не имеет запаха.
¬ытеснением водорода металлами из растворов кислот: Zn + 2HCl = ZnCl2 + H2 ↑.
Ј ислород (ќ2) без запаха и цвета, т€желее воздуха, мало растворим в воде.
1.–азложением перманганата кали€: 2KMnO4 = K2MnO4 + MnO2 + ќ2 ↑;
2.–азложением пероксида водорода:2H2O2 = 2Ќ2ќ + ќ2 ↑.
¬спыхивание тлеющей лучинки, внесенной в сосуд с кислородом.
Ј јммиак (NЌ3) имеет резкий характерный запах, без цвета, хорошо растворим в воде, легче воздуха.
1.¬ промышленности:
3H2 + N2 = 2NH3;
2.¬ лаборатории:
NH4Cl + NaOH = NaCl + H2O + NH3↑.
ѕ–ј “»„≈— јя „ј—“№
| ’од работы | –исунки, реакции, наблюдени€. | ¬ыводы |
| †ќпыт є1 ѕќЋ”„≈Ќ»≈ јћћ»ј ј » ≈√ќ ќЅЌј–”∆≈Ќ»≈. | ||
—оберем прибор.
 ¬ фарфоровой чашке измельчим 1г. NH4CL и 0,5г Ca(OH)2.—месь поместим в сухую пробирку и нагреем. ќсторожно понюхаем полученный газ. ѕоднесем к отверстию трубки лакмусовую бумажку. —оберем газ в пробирку и опустим в нее стекл€нную палочку смоченную в сол€ной кислоте. ¬ фарфоровой чашке измельчим 1г. NH4CL и 0,5г Ca(OH)2.—месь поместим в сухую пробирку и нагреем. ќсторожно понюхаем полученный газ. ѕоднесем к отверстию трубки лакмусовую бумажку. —оберем газ в пробирку и опустим в нее стекл€нную палочку смоченную в сол€ной кислоте.
| «апишем –»ќ: † †NH4CL + Ca(OH)2→ Е..ЕЕЕЕЕЕ ..................................................................... ƒопишем реакцию: NH3+ HCL→ Е..ЕЕЕЕЕЕЕЕЕ... Ќаблюдали: ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ. | ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ |
| ќпыт є2 ѕќЋ”„≈Ќ»≈ »—Ћќ–ќƒј †» ≈√ќ ќЅЌј–”∆≈Ќ»≈. | ||
| ¬ пробирку внесем 1-2 мл. KMnO4 добавим 1мл.сол€ной кислоты. ¬ полученный газ опустим зажженную лучину. |  Ќаблюдали:
ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ.
2KMnO4 = K2MnO4 + MnO2 + ќ2 ↑; Ќаблюдали:
ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ.
2KMnO4 = K2MnO4 + MnO2 + ќ2 ↑;
| ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ |
| ќпыт є 3. ѕќЋ”„≈Ќ»≈ ќ —»ƒј ј«ќ“ј (II) » ≈√ќ ќЅЌј–”∆≈Ќ»≈. | ||
| Ќа дно пробирки поместим 1гр. NaNќ3 (или Nќ3) и добавим 4-5 капель серной кислоты. ƒоведем содержимое пробирки до кипени€. ќсторожно поместите в пробирку медную проволоку. | †ƒопишем реакции и подберем коэффициенты:
NaNќ3+ Ќ2SO4ЃЕЕЕЕЕЕЕЕЕ
†HNќ3(кон.)+—иЃ—и(Nќ3)2+NO2+H2 O
 Ќаблюдали:
ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ.......... Ќаблюдали:
ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ..........
| †ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ |
| ќпыт є4 ѕќЋ”„≈Ќ»≈ ”√Ћ≈ »—Ћќ√ќ √ј«ј †» ≈√ќ ќЅЌј–”∆≈Ќ»≈. | ||
| ¬ пробирку внесем 1-2 кусочка мрамора и добавим 1мл.сол€ной кислоты. ¬ полученный газ опустим зажженную лучину. |  Ќаблюдали:
ЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ.ЕЕ
ƒопишем реакцию (–»ќ):
—а—ќ3 +HCLЃЕЕЕЕЕЕЕЕ
ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ Ќаблюдали:
ЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ.ЕЕ
ƒопишем реакцию (–»ќ):
—а—ќ3 +HCLЃЕЕЕЕЕЕЕЕ
ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ
| ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ ЕЕЕЕЕЕЕЕЕЕ |
ќбщий вывод:...........................................................................................................................................................
...............................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................................
ЋјЅќ–ј“ќ–Ќјя –јЅќ“ј є9
“ема: "»зготовление моделей молекул углеводородов. ќбнаружение углерода в органических соединени€хї
÷ель: »зучить строение молекул метана, этилена, ацетилена, циклопрпана, бензола и их гомологов. Ќаучитьс€: изготавливать шаростержневые модели этих молекул и определ€ть характер св€зей; обнаруживать углерод в органических соединени€х с помощью реакции горени€.
“≈ќ–≈“»„≈— »… ќЅ«ќ–
| 1. ѕри перекрывании всех 4х †орбиталей 2S1, 2Px1, 2Py1, 2Pz1 атом углерода переходит в S р3 Ц гибридное состо€нии. Ќа образовавшихс€ новых 4х †орбитал€х, находитс€ по 1 неспаренному электрону, а сами они направлены к несимметричными вершинам тетраэдра, образу€ валентный угол j = 109028¢ | ”глерод в Sр3 гибридном состо€нии образует -†††††† простую, одинарную св€зь
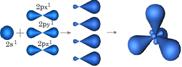 †††††††††††††††††††††††
S р3 Ц гибридное состо€нии. †††††††††††††††††††††††
S р3 Ц гибридное состо€нии.
|
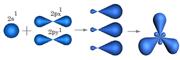
| 2. ѕри перекрывании орбиталей 2S1, 2Px1, 2Py1 атом углерода переходит в S р2 Ц гибридное состо€нии. ѕерекрыва€сь,три орбитали измен€ют форму и станов€тс€ несимметричными, образу€ между собой валентный угол j = 1200. ќрбиталь 2Pz1 остаетс€ без изменений, так как в гибридизации не участвует. | ”глерод в Sр2 гибридном состо€нии образует Ђ=ї двойную св€зь.
 S р2 Ц гибридное состо€нии.
S р2 Ц гибридное состо€нии.
|
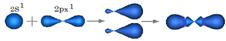
| 3. ѕри перекрывании орбиталей 2S1, 2Px1 †атомуглерода переходит в S р Ц гибридное состо€нии. ѕерекрыва€сь, две орбитали измен€ют форму и станов€тс€ несимметричными, образу€ между собой валентный угол j = 1800.ќрбитали 2Py1 и 2Pz1 остаютс€ без изменений, в гибридизации не участвуют. | ”глерод в Sр -гибридном состо€нии образует Ђ≡ї тройную св€зь
 S р †Ц гибридное состо€нии.
S р †Ц гибридное состо€нии.
|
σ- св€зь (сигма-св€зь) Ч ковалентна€ св€зь, образующа€с€ перекрыванием электронных облаков Ђпо осевой линииї. π-св€зь (пи-св€зь) -это св€зь, образованна€ по обе стороны от линии соедин€ющей центры атомов
ѕ–ј “»„≈— јя „ј—“№
| ’од работы | –исунки, реакции, наблюдени€. | ¬ыводы | ||||||||||||
| ќпыт є1 ќЅЌј–”∆≈Ќ»≈ ”√Ћ≈–ќƒј » ¬ќƒќ–ќƒј ¬ √≈ —јЌ≈ » ≈–ј—»Ќ≈ | ||||||||||||||
| ЌальЄм немного керасина и гексана, подожжЄм. ќбратим внимание на цвет пламени и характер горени€ керасина и гексана | Ќаблюдали: ЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕЕ”рав
ƒата добавлени€: 2018-10-14; ћы поможем в написании ваших работ!; просмотров: 667 | Ќарушение авторских прав ѕоиск на сайте: Ћучшие изречени€: |
|||||||||||||
√ен: 0.308 с.





 Ќаблюдали:
Ќаблюдали:

 .......................................................................
.......................................................................